Retrolental Fibroplasia: A Modern Parable – Chapter 9
Chapter 9
The Determinative Era of Oxygen Treatment
The most recent epoch of opinion/practice concerning oxygen therapy began hesitantly in the 1960s. It slowly became apparent that the old fears of experienced nurses in the early 1950s had not been entirely groundless. The use of oxygen in the treatment of premature infants with obvious signs of oxygen deficiency posed a classic Scylla-and-Charybdis-like dilemma for physicians. Liberal administration. was thought to increase the risk of RLF, whereas restricted use was expected to increase mortality and the likelihood of brain damage in survivors. Repeated determinations of the state of oxygenation of each sick infant seemed the obvious approach to guide treatment and avoid impalement on this two-horned dilemma in the nursery.
The monitoring of treatment by direct ophthalmoscopy to detect oxygen-induced narrowing of retinal arteries proved to be impractical at a time when information was needed most. Examination of desperately ill infants in the first hours and days of life was difficult for the examiner and dangerously exhausting for the patients. Moreover, visualization of the retina was often blurred by vitreal haze.
The Nuffield Neonatal Research Unit at the Hammersmith Hospital in London proposed a plan, the pure oxygen breathing test (Table 9-1), which gained fairly wide acceptance. This scheme formalized a trend already underway, a shift of attention from the concentration of oxygen it inspired air to the measurement of oxygen tension in serial samples of arterial blood (usually from the aorta via an umbilical artery catheter). The question of whether there was a threshold arterial condition (elevated oxygen tension over a period of time) for RLF to develop was as difficult to answer as the earlier poser when oxygen was measured in inspired air only Although it seemed reasonable to attempt to control blood oxygen tensior at the levels found in “normal” infants breathing room air (about 60-100 mm Hg), it would have been surprising if this eliminated RLF completely since, as I have already indicated, there were documented examples of the disorder in infants who had never received supplemental oxygen. Indeed the Hammersmith group encountered RLF in an infant whose oxyger tension (in serial samples of arterial blood) never measured more than 80 mm Hg. The unanswered question remained, Will the new “determinative’ policy be associated with an increased frequency of RLF?
Table 9-1
Breathing Pure Oxygen: A Test To Determine
Oxygen Treatment In Respiratory Distress Syndrome
| Arterial Oxygen While Breathing Pure Oxygena | Recommended Change in Oxygen Treatment |
| < 100b | Remain in 90-100% oxygenc |
| 100-150 | Decrease in 10% steps (remeasure) |
| 150-300 | Decrease in 20% steps (remeasure) |
| > 300 | Decrease in 40% steps (remeasure) |
| a Partial pressure of oxygen (in millimeters of mercury) in blood drawn from the aorta through an umbilical artery catheter, after the infant has been breathing pure oxygen by face mask for 15 minutes. | |
| b Asymptomatic infants breathing room air have arterial oxygen measurements ranging from about 60 mm Hg at birth to about 100 mm Hg at 54 hours of age. | |
| c Administered in a Plexiglass box enclosing the infant’s head. | |
There was no national data-pool in the United States from which the ongoing incidence of RLF could be determined. Blind or visually impairec children in a community were usually not reported to service agencies until they appeared for schooling. Thus, there was an agonizing lag of 5 years of more before a rise in frequency could be detected unless the increase was explosive. In this uneasy atmosphere of doubt and concern, I approaches the National Society for the Prevention of Blindness and suggested that a survey be conducted to attempt early detection of any rise in the occurrence of RLF: The year 1967 was chosen as a representative one for the new oxygen practices.
Some 1100 North American hospitals were canvassed by mail with a request for the numbers of infants with RLF born in the survey year. Only 369 of the hospitals responded, and the crude estimate of frequency of occurrence provided no clear answer to the question. The fact that only 33 examples of RLF were uncovered was not reassuring. On the contrary, the results suggested that many infants-at-risk were not being examined in 1967.
Beginning in 1969, five hospitals throughout the United States undertook a forward-seeking, collaborative study to determine the “safe” arterial oxygen tension to reduce the risk of RLF. The goal was not realized. Observations in 719 infants, most of whom were being treated for respiratory distress, indicated that there was no significant difference in average oxygen tension in infants who developed cicatricial RLF and those whose eyes remained normal. In both groups average arterial oxygen values did not exceed the “normal range” (60-100 mm of mercury).
Recent findings have compounded the problem of deciding whether or not the incidence of RLF has been increasing during the late 1960s and 1970s among premature infants (particularly those with moderate or no respiratory symptoms who were exposed to relatively little supplemental oxygen). Improved techniques (retinal photography, indirect ophthalmoscopy, and fluorescein angiography) for examining the retinas of newborn infants have shown that the blood vessel changes of RLF (Fig. 9-1) occur much more often than appreciated in the past. It seems unlikely that the high frequency is explained entirely by latter-day liberal oxygen practices. For example, Baum found many residual changes in the eyes of 52 late-teenagers in Denver, Colorado, who had been born prematurely during the years before and after oxygen restriction. He observed only three persons with normal eyes, 14 had some degree of cicatricial (scarring) RLF, and 35 showed abnormal twists and turns of the retinal arteries (Fig. 9-2).
From these observations the idea has gradually emerged that vascular RLF is a common developmental aberration in infants with immature retinal vessels who breathe air, with or without supplemental oxygen. Moreover, previous preoccupation with the role of oxygen in the production of RLF appears to be giving way to wonder about the complementary observation, i.e., most infants (and all experimental animals) who develop the vascular changes of RLF, even those exposed to high oxygen for prolonged periods, do not go on to develop cicatricial changes which produce blindness or retinal scarring (Fig. 9-3). In this connection, the re-awakened interest in evaluation of a protective effect of vitamin E or a potentiating effect of blood transfusion may lead the way to new investigations. Other factors, yet unexplored, may play a role in resisting the effects of oxygen on immature retinal vessels or promoting uncomplicated regrowth after the retinal net of vessels has been obliterated by high oxygen exposure.
New observations and speculations have raised additional doubts about the quick and simple answers of the 1950s to the complex questions concerning oxygen treatment of premature infants. Early mortality among infants in the years before oxygen restriction, during restriction, and in the recent determinative era was reviewed by Cross in 1973 and Bolton and Cross in 1974. From this perspective (Fig. 9-4), the cost of preventing RLF during the period of oxygen restriction was estimated from the decrease in blindness and the rise of day-of-birth deaths during the two decades after the RLF epidemic was brought under control:
If we say that blindness has fallen from around 50 a year [in England and Wales], and if we say that there has been an excess number of deaths of something over 700 per annum, then it would seem that each sighted baby gained may have cost some 16 deaths.
Although it is impossible to prove the Cross hypothesis, the startling calculation raised the possibility that there is one more blot on the doleful record of the RLF incident. Additionally, the magnitude of “cost” of oxygen restriction in terms of brain damage has not been estimated. For example, it is conceivable that there might be a very small increase in the risk of RLF in contrast to a large reduction in the risk of spastic diplegia (p 56) for infants exposed to continuous supplemental oxygen in concentrations lower than those formally tested in the 1953-54 Cooperative Study. If it should be so, an agonizing reappraisal of the issues involved in oxygen treatment of premature infants would be in order.
The entire matter of long-term outcome in survivors of the RLF epidemic has received relatively little attention from the medical profession. During the years after the RLF epidemic was “over” for the attendants in premature nurseries (and, later, in intensive care units for the newborn), its impact was in full force (Fig. 9-5) for parents, teachers, social workers, mobility instructors, and others concerned with rehabilitation of the lives of these affected persons. Moreover, there were persistent reports from those with considerable past experience that RLF-blinded children and adults were “different” from other congenitally blind persons. The difference in performance centered around spatial orientation, and the dysfunctions were spotty. For example, the director of an orientation center for the blind told of an RLF-blinded man who played Bach fugues on the piano flawlessly, yet he was unable to replace his shaving razor in its case. When he dropped his towel, he reached up to retrieve it, not down. There have been other anecdotes and allegations of RLF-associated abnormalities but they have never been subjected to intensive investigation. Although the studies in the 1950s of prolonged exposure of newborn animals to oxygen failed to disclose impressive evidence of gross or microscopic structural changes in the brain, the subject has not been explored exhaustively. Many metabolic reactions in the body are influenced unfavorably by elevated oxygen tension. There is evidence which suggests that the harmful affects are related to the reduction of small amounts of molecular oxygen to transient superoxide free-radicals within the cells of the body. These highly reactive substances produce irreversible damage to vital chemical compounds within cells (enzyme proteins and membrane lipids). In view of earlier observations on the association between iron treatment and RLF (see p 19), it is interesting to note that inorganic iron has a catalytic action which promotes the generation of the potent free-radicals. The potential importance of these considerations to present-day debates concerning the care of newborn infants can be summarized by one question:
Are the eye complications of excessive oxygen the most sensitive endpoints by which to judge a determinative policy of oxygen treatment of premature infants?

Early changes of RLF as seen with the aid of retinal photography and fluorescein angiography. A sharp silver- or gray-appearing abnormal shelf of tissue has formed a border between the central retina (to the left) which has blood vessels and the peripheral portion (to the right) which is devoid of vessels (in contrast to the very gradual change in appearance between these two areas of the retina in the unaffected premature infant). A fluorescein angiogram of the border structure, seen here, reveals that the ledge is a complex of blood vessels in which there is a direct shunt of blood between the arterial and venous circulations.

Tortuosity of retinal arteries seen on a photograph of the retina in an ex-premature teenager. Note the hairpin bends in many of the arteries, similar to the changes in the acute vascular stages of RLF during the newborn period (Fig. 1-3, A).

Limitations in the relationship between high oxygen concentration and RLF (acute blood vessel changes-dashed circle; scarring and blindness-small closed circle). Most premature infants exposed to supplemental oxygen do not develop RLF. Some infants who develop RLF have never been exposed to supplemental oxygen.

In the U.S., England and Wales, day-of-birth death rates depart from the projected exponential curves (dashed lines) in the 1950s. The observed death rates began to fall in the mid-1960s but remained displaced from the “expected” line. B. By contrast, death rates in England and Wales for days-of-life 1-6 show a smooth exponential decline from 1935 through 1970. (See chapter notes for further comments.)
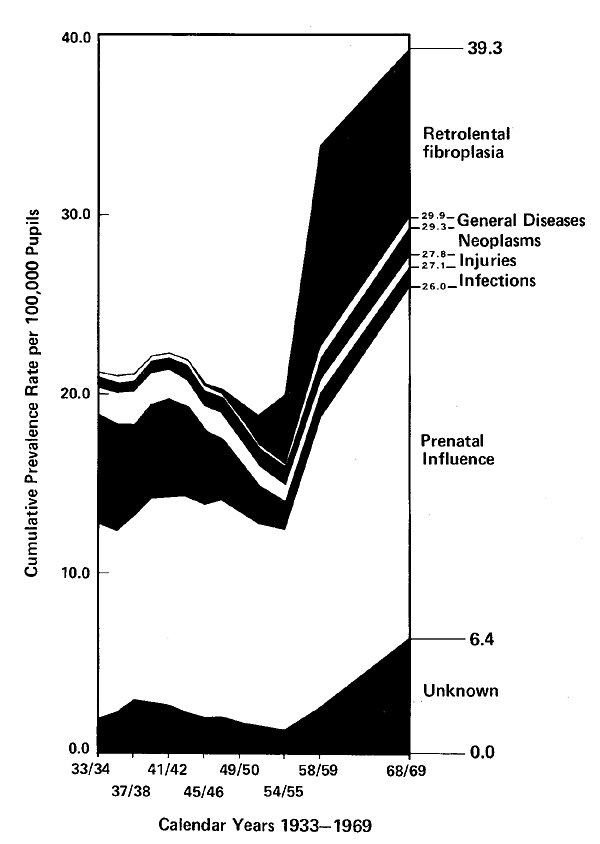
Estimated blindness prevalence rates (cumulative) in school-children by cause in the school years 1933-1934 to 1968-1969.
Last Updated on 02/28/24